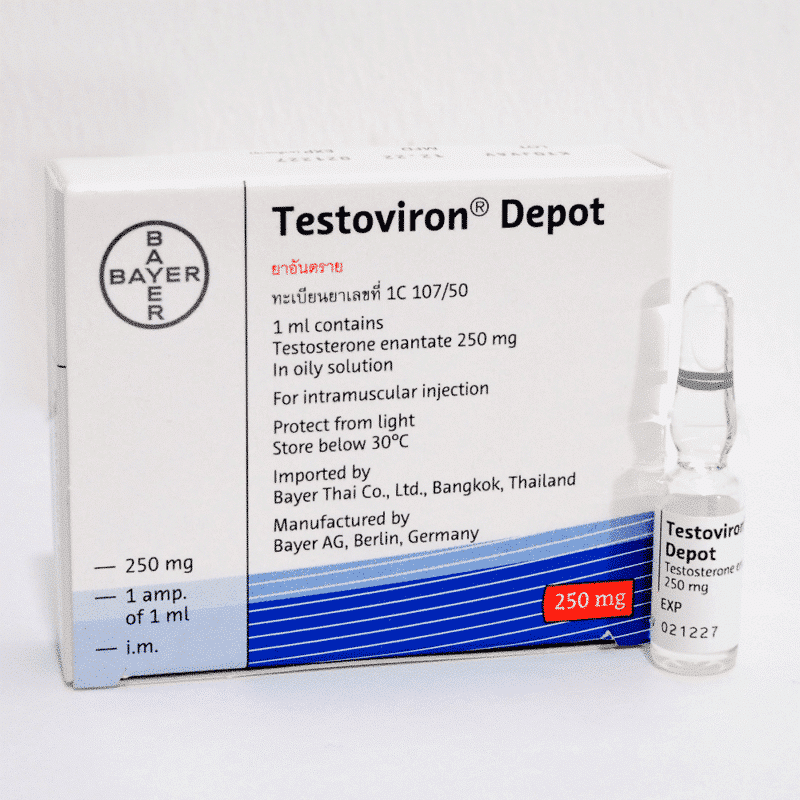
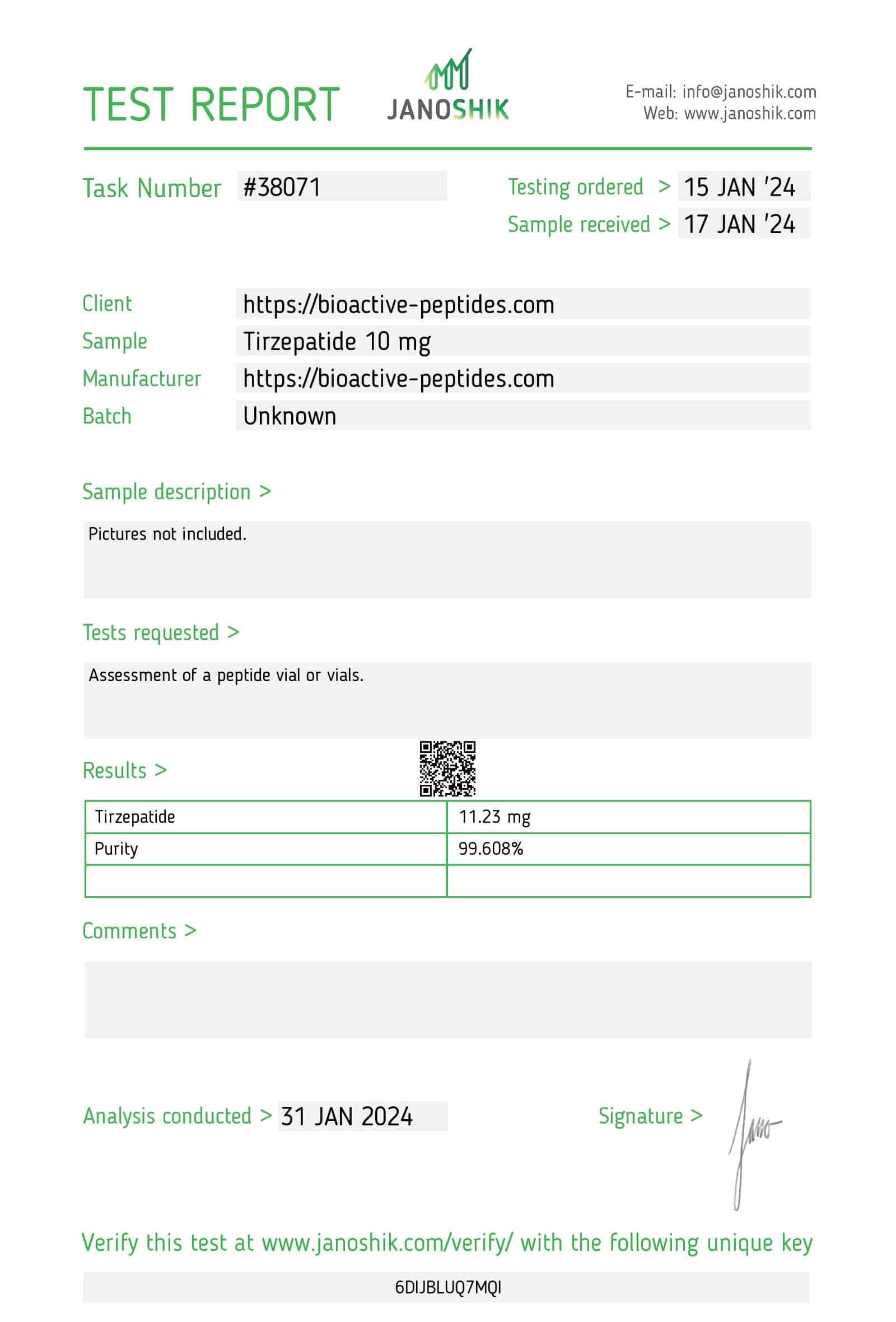
Tirzepatide 10mg
Tirzepatide is a synthetic derivative of gastric inhibitory polypeptide (GIP) that has simultaneous glucagon-like peptide-1 (GLP-1) functionality as well. This combination allows Tirzepatide to lower blood glucose levels, increase insulin sensitivity, boost feelings of satiety, and accelerate weight loss. Tirzepatide was developed to fight type 2 diabetes, but has additionally been shown to protect the cardiovascular system and act as a potent weight loss agent.
Properties
- Chemical Formula: C225H348N48O68
- Molecular Mass: 4813
- Synonyms: Tirzepatide (LY3298176) 2023788-19-2
- PubChem: 156588324
- Total Amount of the Active Ingredient: 10mg (1 vial)
- Shelf Life: 36 months
Benefit
TIRZEPATIDE ACTION
Tirzepatide is a GIP-analog that appears to activate both the GLP-1 and GIP receptors. Generally, the peptide is reported to be a more potent activator for GIP than GLP-1 receptors. Both of those receptors can be found in the pancreas but also in other organs. Usually, the small intestine produces GLP-1 and GIP when certain nutrients are in its lumen. For example, researchers posit that GIP is triggered by the hyperosmolarity of glucose that enters the duodenum after a meal. The GIP and GLP-1 peptides are then released in the blood and travel to the corresponding receptors. Activating either of those receptors in the pancreas may stimulate the beta-cells to produce insulin in a glucose-dependent manner. However, the effect is glucose-dependent, so GIP or GIP analogs like Tirzepatide do not appear to trigger insulin secretion if glucose levels are low or average, and this reduces the risk of hypoglycemia. GIP receptors can also be found in the gut, adipose tissue, heart, pituitary, and inner layers of the adrenal cortex. GLP-1 receptors have also been discovered in the gut, exocrine pancreas, brain, heart, lung, and kidney. Thus, Tirzepatide may have benefits other than stimulating insulin secretion, and scientists are currently exploring its potential for a wide range of conditions.
TIRZEPATIDE AND GLYCEMIC CONTROL
The main potential benefit of Tirzepatide is the reduction of blood sugar levels and improved glucose control, as exhibited in test subjects with type 2 diabetes. Studies suggest that due to this dual agonist behavior, Tirzepatide may have a high efficacy for improving glycemic control compared to other anti-diabetic medications such as GLP-1 agonists, SGLT-2 inhibitors, or DPP-4 inhibitors. Moreover, Tirzepatide may also help improve insulin sensitivity and beta-cell function in subjects with type 2 diabetes. Studies note that the peptide appeared to reduce insulin resistance as measured by the HOMA2-IR index, and the effect was more remarkable when compared to GLP-1 agonists. Thomas et al. explained that the improvement appeared to be primarily independent of body weight reduction and noted that “weight loss explained only 13% and 21% of improvement in HOMA2-IR with Tirzepatide.”
WEIGHT LOSS
A meta-analysis of 9 studies that covered more than 7,000 test subjects with diabetes reported that Tirzepatide may lead to more weight loss than GLP-1 agonists such as Semaglutide. The duration of the studies ranged from 8 to 52 weeks, and the subjects achieved about 5kg weight loss on average. Moreover, Tirzepatide appeared to exhibit better glycemic control than Semaglutide and insulin. Permana et al. concluded, “Tirzepatide has shown superiority in glycemic control and body weight reduction with a good safety profile in patients with T2D.” Tirzepatide has been suggested to induce weight loss regardless of whether the test subject has diabetes. In one trial with 2,539 obese subjects and at least one weight-related complication (excluding diabetes),Tirzepatide therapy was reported to lead to a dramatic reduction in body weight. In 72 weeks of therapy, subjects had at least a 20% reduction in body weight or more, compared to only 3% weight loss in the placebo group for the same period.
HEPATOPROTECTION
Tirzepatide has been reported to improve markers of liver function in test subjects with non-alcoholic fatty liver disease (NAFLD). Apart from reducing liver enzymes such as ALT and AST, the peptide also appeared to lower markers of liver cell death, such as K-18, and markers of liver fibrosis, such as Pro-C3.[10] The results of another trial suggests that Tirzepatide may also lead to a significant reduction in liver fat content, which is another direct marker for NAFLD. In 52 weeks, the peptide appeared to lead to an average 8% reduction in liver fat.
Dosage
Doses start at 2.5 mg once weekly; after 4 weeks on the 2.5 mg dose, increase to the 5 mg dose once weekly. If higher doses are required, increase by 2.5 mg increments after at least 4 weeks at the current dose. The maximum dose is 15 mg once a week.
Disclaimer
The information provided above is not intended to substitute medical advice, diagnosis, or treatment. Should you have any questions regarding a medical condition, seek the advice of your physician or a qualified healthcare provider. In no case should medical advice be disregarded or delayed because of what you have read or seen. We bear no responsibility or liability for your use of any of our research compounds and products. Please note that they are being sold for research purposes ONLY. We do NOT condone any personal use.























รีวิว
ยังไม่มีบทวิจารณ์