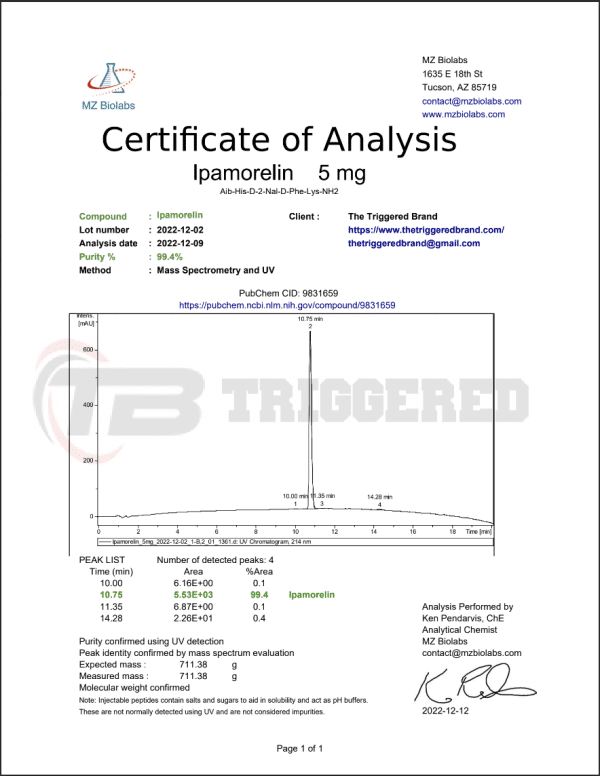
Ipamorelin 5mg
Ipamorelin เป็น pentapeptide ที่ประกอบไปด้วยอะมิโน 5 สาย ทำหน้าที่เป็นเหมือนยาที่ช่วยจับ receptor ของเซลล์และกระตุ้นให้เกิดตอบสนองและการหลั่ง Growth Hormone ระบบการทำงานของ Ipamorelin นั้นจะช่วยให้เปปไทด์กระตุ้นการหลั่งฮอร์โมนที่ต่อมใต้สมอง และในขณะเดียวกันในงานวิจัยพบว่าเปปไทด์สามารถกระตุ้นการหลั่ง ฮอร์โมนโซมาโทสแททิน Ipamorelin ยังช่วยกระตุ้นการผลิต IGF-1 ซึ่งมีความสำคัญในการกระตุ้นและซ่อมแซมเนื้อเยื่อกล้ามเนื้อและกล้ามเนื้อลาย
คุณสมบัติ:
สูตรเคมี – C38H49N9O5 xC2H4O2
มวลโมเลกุล: 711.9
ชื่อสินค้าประเภทเดียวกัน: 2-Methylalanyl-L-histidyl-3-(2-naphthyl)-D-alanyl-D-phenylalanyl-L-lysinamide, 170851-70-4, C38H49N9O5, CHEMBL58547, SCHEMBL183305, SCHEMBL8169698, MolPort-039-139-953, 5020AF, ZINC29562299, AKOS030573449, DB12370, NCGC00167295-01, FT-0696037
สายพันธุกรรม: Aib-His-D-2-Nal-D-Phe-Lys-NH2
CAS Number: 170851-70-4
PubChem: CID 20754357
ปริมาณ Active Ingredient: 5 mg (1 vial)
อายุการเก็บรักษา: 36 เดือน
Dosage
Based on user experience, for fitness goals, you can inject 200mcg-300mcg of ipamorelin up to 3 times a day. For anti-aging purposes, use it once a day with approximately 200mcg of ipamorelin.
Disclaimer
The information provided above is not intended to substitute medical advice, diagnosis, or treatment. Should you have any questions regarding a medical condition, seek the advice of your physician or a qualified healthcare provider. In no case should medical advice be disregarded or delayed because of what you have read or seen. We bear no responsibility or liability for your use of any of our research compounds and products. Please note that they are being sold for research purposes ONLY. We do NOT condone any personal use.

























รีวิว
ยังไม่มีบทวิจารณ์